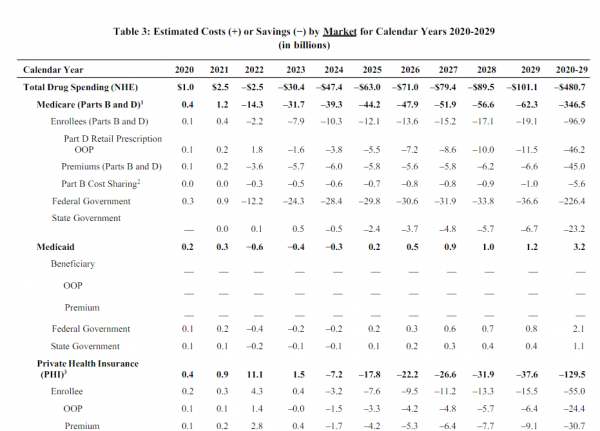
The Congressional Budget Office released a partial analysis of HR3, the House drug pricing bill. The headline numbers are big:
$480 billion dollars in lower drug spending over a decade is real money even in the context of federal health care spending.
However there is an innovation/introduction of new treatment cost:
its preliminary estimate is that a reduction in revenues of $0.5 trillion to $1 trillion would lead to a reduction of approximately 8 to 15 new drugs coming to market over the next 10 years. (The Food and Drug Administration approves, on average, about 30 new drugs annually, suggesting that about 300 drugs might be approved over the next 10 years.) The overall effect on the health of families in the United States that would stem from increased use of prescription drugs but decreased availability of new drugs is unclear.
This is the trade-off that economists worry about. What is the loss in innovation for cost savings?
The CBO does not project what type of new drugs that would have been produced won’t be produced. If those never approved counter-factual drugs are merely drugs that “cure” bushy eyebrows or are the seventeenth and eighteenth variants of a general use class of medications that have no specific targeting features, the loss of innovation is fairly low. If the lost counter-factual drugs are something that has the same impact as the (expensive) Hep-C cures, then the loss of innovation is significant.
There are very few free lunches. Almost everything has trade-offs. If we can get some more information on what types of drugs would not be developed/approved, that would be extremely valuable. Without that information, we instead need to assess best case, probable case and worse case scenarios to decide what types of trade-offs are acceptable for spending less on pharmacy. Half a trillion dollars over a decade is a real benefit, so figuring out what the costs are likely to be is a critical step to determine if that benefit is good enough.

planetjanet
Just wow. I did not know this legislation was being developed. This has felt like the holy grail since the Bush era. Allowing the government to bargain with industry on drug prices for Medicare/Medicaid is an obvious action we should take. But this extends the negotiated drug prices to private insurance as well. So you avoid punishing some people to help others. I love it.
patrick II
Take half of the $480 billion gained and do government research instead of private research, and look for real gains in new drugs. Too many drugs or drug systems researched by private corporations, like the epinephrine injector I use, earn a patent for being “new” when some minor change has been made. Make the incentive for drug research solving an important problem, not just have the incentive be profit, not any new or real gain. I have read too often about drugs that were not being researched because their potential use would not be profitable.
How does the change in law impact situations like Shkreli?
Cheryl Rofer
This is a great piece of analysis.
The tradeoff is obvious. Eight to fifteen drugs out of 300, many of which are of no use to the consumer, versus $480 billion savings?
justawriter
Ban TV prescription drug commercials and tell the companies they can double their research spending with the money they save. (Comment is for satirical use only. Please consult with your lobbyist to see if satire is right for you.)
Mousebumples
@Cheryl Rofer: I certainly can’t say for sure, but as a pharmacist, I suspect the “me too” type drugs (eg slight variations on existing meds without much added benefit) would continue to be introduced since they cost less to create as less innovation is needed.
I agree with Patrick II that using some of the savings to sponsor government research is a good idea. Living in Wisconsin, when I used to work for Walgreens, I’d tell new warfarin patients about how the WARF in the drug name came from the Wisconsin Alumni Research Foundation.
Typically, after a new drug product is found by the NIH, I think they give or sell it to a company to produce. I wonder if it might be worth reconsidering that approach – if only to cut costs with the final drug products. Though, I admit I don’t know enough about the “next steps” after a new drug product is identified.
Rusty
With a position that is tangent to the drug discovery field, I would very like to see the analysis and what data (and how old) was used to generate this conclusion. The drug companies have cut back significantly on drug discovery, the timelines are very long and stock price is based on the last quarter. It is cheaper and easier to let the government fund research, and go after that which looks promising. Even there, the vehicle is much less taking on the cost of further development by acquiring or licensing in-house, but by using various start up vehicles with seed money from many sources and the ability to cut losses quickly without putting anything on the books. With this the new reality, taking the savings and rolling some back into government funded research makes more sense. I suspect the “loss” of new drugs is less likely given the new realities of drug discovery.
grelican
I’m in health policy myself, currently exiled from government. One argument I’ve actually liked to hear out of Trump’s HHS is that other nations are free-riding and/or being subsidized by our high prices. Another way of framing it is that if we don’t negotiate for lower prices while everyone else does, then we’re suckers. I think that’s why this issue triggers Trump at a visceral level. I don’t see it in such negative terms, but it’s true that our outsized prices here mean others don’t have to pay the same amount to ensure the rate of drug discovery (and profit) that these multinational corporations accrue. I just don’t think these companies will wither away if that changed.
Here’s why I think those arguments are bunk. Look at the CBO’s projections of cuts to innovation as a result of HR 3. The study parameters are highly limited. They don’t account for secondary effects as the global market adapts. These companies are multinationals, with the US being just one of many lucrative markets. If we did what other countries do, then the companies would come to the negotiating table in the UK or France with a different set of calculations, and demand more money from them. Like so many things, we act like the US is a planet unto itself, and if we pay less for drugs, then the whole thing falls apart. These companies are global. Making money would be a little harder, and their margins would likely shrink at least in the short term, but there’s no real reason to think that innovation would dry up just because the United States stops paying whatever price they demand.
What I mean is that other countries don’t just unilaterally declare that they will only pay $100 for a drug that we pay $1000 for, and then that’s it. They do it based on its relative value, and they pay handsomely for innovative products. They require life sciences companies to open their books, break down what their R&D, overhead, and profits look like, and then they set a price that rewards them for making something that helps people. If the facts change, so do the prices. What they don’t do is let themselves get ripped off by diluting their nation’s negotiation power, or buying things that are little better in terms of marginal outcomes improvements.
I’ve long struggled with the innovation vs. cost dilemma. The hard thing to accept is that pharma companies are bloodthirsty capitalists who are also curing cancer. There’s a lot of moral ambiguity there, but one thing I’m reasonably sure of is that if we drove a harder bargain for the US patient, the bloodthirsty capitalists will find a way to keep doing what they’re doing and turn a profit. Capitalists always act like invincible juggernauts of progress, going toe to toe against all manner of challenge and winning the day. Then the regulators show up and they’re a bunch of abused puppies. The problem is really political. This is an industry where $100m of lobbying can easily translate to $100b of profit, so we hear a lot about how we have no choice and it’s either granny’s cancer pills or leaches, and nothing changes.
Roger Moore
@patrick II:
Or don’t spend it on drugs at all, and instead spend it on subsidizing care for people who currently have trouble getting it. Drugs are only one of many kinds of medical treatment. Our society loves them because A) they’re sciency, B) they let the doctor offer something quickly and move on to the next patient, and C) they create concentrated economic interests that have lots of money to lobby the government (and propagandize the public) to protect their business model. I think we’re putting too much time and effort into drugs over other forms of treatment, and we would still be focusing too much on drugs even if we were paying as little for them as other countries are.
RSA
I’m wondering about what retrospective analyses show. For example, sample ten year periods from the past, and within each period sample a 90% proportion; compute some measure of loss. Lots of assumptions would be needed, but maybe it could be informative?
Another Scott
@grelican: +1
I’ve been surprised how many meds in our household are generics from India and Canada and elsewhere these days. The market is moving, whether the US wanna-be monopolists like it or not. They can help guide the way those moves happen, or they can continue to try to bribe their way (via lobbying) to their preferred outcome. But the market is going to change. It’s not sustainable.
“If something cannot go on forever, it will stop.” – Herbert Stein.
Cheers,
Scott.
jl
Thanks for very interesting link and analysis. As I’ve noted before, the US has pushed IP and patent protection in the direction of corporate and patent holder advantage to historical extremes, both in the US and around the world. There are alternatives to current patent approach for drug innovation with a high public need.
The Roles Of Patents And Research And Development Incentives In Biopharmaceutical Innovation
https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2014.1047
The US patent system is a policy not a fact of nature. The US patent system could be altered to prevent excess rents accruing to blockbuster drugs, and excessive rent seeking. Changing the profile of annual fees to keep the patent in force is one example to follow practice in the rest of the world, is just one example.
The annual number of new molecular entities patented as been at a plateau since late 1990s, so unclear whether current trend in prices is doing anything for innovation. A lot of recent patent rent seeking in US drug patents is bundling of active drug and drug delivery devices, something that is decoupled in most of the rest of the world. Has made wider access to new devices for a wide variety of drugs in rest of world.
MattF
However, predicting income from new drugs is not so easy:
https://blogs.sciencemag.org/pipeline/archives/2019/07/12/revenues-from-new-drugs-and-thoughts-on-chance
Amir Khalid
@justawriter:
Why do American drugmakers get to advertise prescription drugs to the public? Is it a 1st Amendment thing?
MattF
@Amir Khalid: No. Advertising prescription drugs in non-medical media used to be forbidden. Not sure when that changed.
patrick II
@Roger Moore:
I guess putting a dollar figure on it is the wrong framework for better choices. But there is opportunity for beneficial research that has been passed by. I will cite specifically the vaccine for the ebola virus which was discovered, tested ou monkeys and sat on a shelf for 10 years without further testing because of no immediate potential for profits. Then an Ebola outbreak happened, thousands died, and no vaccine.
Or, there are many untested natural, unpatentable products that might prove very inexpensive. Too inexpensive really. Perhaps I have just been propagandized too much by GNC, but Cheap, natural ‘drugs’ might be worth a look.
Yeah, there are a million ways to spend money to improve health – – better, healthier lunches for poor school children would be a start. But I think somewhat more profit independent drug research might be helpful too.
jl
As an example, though this is my anecdata from physicians and pharmacists who have some understanding of patent economics and comparative international treatment access and prices, the role of device/drug coupling in patents has been particularly important in insulin in the US. Insulin prices have been in the news.
Again, the design of a patent system is a policy choice not a fact of nature. And a patent system has many dimensions and many leverage points for fine tuning, particularly in controlling rent seeking. There are significant differences around the world.
We also need to look at approval and licensing process in the US. What are incentives for US patent office to give patents for me-too and knock-offs, and for FDA to process approvals for new patented drugs, versus approvals and licensing for existing drugs, patented and generic. Some of the horror stories in drug price increases have more to do with time lag for approvals and licensing than patents themselves.
grelican
@Another Scott: The market’s actually doing a decent job of pricing drugs once they go generic. We aren’t nearly as bad of an international outlier in that regard, and sometimes we actually come out ahead. Nevertheless, there are real problems in the generic market. The brand companies fight tooth and nail to keep their patents because they know their profits will be competed away pretty effectively with a little competition. Good news is that there’s a reasonable consensus that we need a more functioning market for generics. It’s addressed in the House and the Senate legislation.
My view is that the most pressing and politically difficult issue is for new products where competition does not, and perhaps cannot exist– so-called “single source” drugs. Companies do need to be rewarded for innovation, and to bring these quite amazing products to market. Every regulator around the world accepts that premise. It’s just that most of the world doesn’t allow these companies to seek a disproportionate amount of the rewards on the backs of their citizens. That’s precisely what HR 3 targets, and that’s what has to change. There’s simply no market mechanism to design around. One way or another, we need to cap these prices.
PAM Dirac
@patrick II:
Most serious estimates of what it would take to completely replace private pharma with government research are in the range of $50-75 billion per year, so the $480 billion prob wouldn’t cover a complete replacement. You could think of a hybrid setup where the government just takes on the most risky efforts and the orphan diseases and that amount of money might provide a pretty reasonable support for that effort. On the other hand the most risky efforts tend to be the most expensive, so it might not change things much.
Carol
My neighbor, who is a retired pediatric oncologist, claims that universities are the ones that do most of the research and that pharma purchases it from them. First, drug company marketing, especially on tv, should not be an operating cost that’s added to the price of the drug, and second, the price of a drug should depend on the actual research a company does, and third, patents should be shortened. That’s my opinion.
BigJimSlade
I’ve got a question I’ve been curious about for a while: how much of the innovation in drugs and medical procedures has been coming from America vs. Europe (and the rest of the world)?
On a semi-related note, I think just over a decade ago a friend of a friend joined us for brunch. He worked for a company that was trying to improve imaging – mostly software, I believe. He said he loves working with Americans because the people (doctors) in charge of the decisions were at the top of their field and really interested in innovation. In the UK he said the people he dealt with were just, you know, somebody with a job. So I’m always concerned about how much we want to turn our health care system, however much I think it’s absurd, into a bureaucracy.
PAM Dirac
@Carol:
There are a lot of claims along these lines but the only attempts I’ve seen to actually total it up paint a very different picture. For example see here for a discussion of one such count that came up with a figure of 15-20% for the years 1998-2007. I have also not seen any comparison that shows drugs developed in academia are any cheaper than pharma developed drugs. Because of Bayh-Dole institutions funded by the government have the right to patent and manage any discoveries they make. My personal experience is that universities are not at all shy about grabbing as much as they can for anything that comes out of their labs.
jl
@Carol: Some complications to the issues you raise
” the price of a drug should depend on the actual research a company does”
There are two dimensions to getting paid for the research done. The first is who gets the patent from US patent office, and how the design of the patent system control rent seeking and excess profits. The second is who gets the approval from FDA. A large amount of the research for approval is in phase III trials. Often, whoever has the financial means to get a phase III trial done first determines who gets the FDA approval. There is a lot of sophisticated gaming during development on timing of US patent office patent approval and FDA approval to try to lock in excess profits from both. Often joint public and private basic research partnerships engage in this gaming. Phase III trials are not by any means innovative research. They need a minimum competence at doing randomized controlled trials, and they are expensive. The rights to do a phase III trial could be auctioned off in some circumstances.
“and third, patents should be shortened”
Many patents are effectively shortened. The timeline of patent protection now usually means that the more time spent and more carefully the pre-marketing research is done, the less time on post-marketing patent protection. This arrangement produces perverse incentives to rush the pre-marketing research in order to lengthen the effective post-marketing patent protection. The criticism of the US FDA in the 1970s was that it was inefficient, and reform could produce both more safety and more innovation. It is very unclear that the current US drug R&D, patent and FDA approval system has delivered on that promise.
Also, there is a problem with whether current US drug patent and approval system fulfills the historic social bargain inherent in the idea of a patent. Traditionally, the tradeoff is that company gets monopoly rights for production and marketing during patent period, but after the period, all knowledge required to produce and use the product is made public. Right now drug companies don’t have to disclose much. More through disclosure of what drug companies know about their drugs could solve a lot of problems. And there is a lot of experience in dealing with this problem, which has been around for 150 years at least. Big problem with 19th century chemical industry patents. So, there is knowledge base for sound policy making on it.
Roger Moore
@patrick II:
Part of the problem is that even if the drug itself is cheap to produce, the approval process can be expensive. Running a series of clinical trials to prove that your drug candidate is actually beneficial is expensive, and there’s no guarantee of a payoff because there’s a substantial chance that it will fail along the way. There’s certainly room for improvement, e.g. by having government funded clinical trials for orphan drugs, but the truth remains that drug development is inherently expensive.
Another Scott
@Carol: It’s probably hard to say where the university drug research money comes from. Lots of university research is becoming more “business” focussed. “User-facilities” that any company can purchase time at; grants to researchers to fund something that companies are interested in; endowed chairs funded by corporations; etc. The days of state and federal governments funding pure, no holds barred, university research are long gone. Yes, the NSF and various other parts of government still fund a lot of research at universities, but it’s not enough because of cuts in other higher education funding.
Cheers,
Scott.
PAM Dirac
@Roger Moore:
More than substantial. Only about 10% of drugs that make it to clinical trials actually are approved. It is only 5% for cancer drugs. The vast majority of the failures are not at all controversial; the trials show them to be too toxic or not to have any efficacy. I remember about 20 years ago a company that made a shark cartilage preparation wanted to do a clinical trial with their product. They were serious about doing the trial right and a number of my NCI colleagues were quite impressed with the plan they put together. For all their good intentions and work, the final result was that they spent $40-50 million of the company’s money to prove their product was worthless. Slapping that nudge, nudge, wink, wink disclaimer on a product is massively more profitable.
patrick II
@Roger Moore:
patrick II:
Sorry, Roger, that is my poor writing skill. What I meant was they would be unpatentable and sold at too inexpensive a price to make the expensive testing investment worthwhile. I know the testing is expensive, but if the payoff, not measured in terms of profit but of increased public health, is worth it, that is what good government is for.
patrick II
@PAM Dirac:
I read Roger to imply, perhaps wrongly, that most private drug research would continue. But as discussed, too much current private drug research is to tweak some existing medicine so the patent can be extended and profits continue, not for any large public health benefit. So, you wouldn’t need all of that money, and hopefully, public research would be based on potential public health benefits, not just profits.
Roger Moore
@Carol:
This may be true, but it’s deeply misleading. Universities do almost all the basic research, but there’s a huge amount of applied research that goes into converting a new scientific study into a usable drug, and that applied research takes place predominantly in industry. Not all that applied research is in big pharma, though. There is a whole layer of startups, often founded by university researchers who want to commercialize the discoveries they made in the university, that does a lot of the early phase development. When they have something promising, big pharma buys them out and puts their money and expertise into converting their drug prospect into a drug product.
I agree that direct to consumer marketing should be drastically curtailed, if not completely banned. And we should do more to rein in drug prices. But in a lot of cases there are serious problems with patent protection being too short rather than too long. It takes a long time to go from when a company applies for a patent on their new drug to when that drug actually hits the market, and that development time severely eats into the value of the patent.
What’s really wrong is the way drug companies can extend the lifetime of their patent protection by filing new claims around an existing active ingredient. So first they patent the active ingredient (e.g. aspirin). Then it takes them a long time to go through the development phase and clinical trials, so there might be only a few years left on their original patent by the time they’re finally allowed to sell it. But in the mean time, they’ve been developing new formulations (i.e. aspirin mixed with other ingredients to make an extended release version) and patenting those. Because the drug companies actually sell the formulated product and not just the active ingredient, and because there’s a patent principle called the doctrine of equivalents that allows a company to patent not just their extended release formula but any other formulation that has similar pharmacokinetics, they’re covered for much longer than the original patent. If they keep developing new formulations, they can keep competitors off the market for a long time after the initial patent has expired. And then they can do stuff that really ought to be banned by the FTC, like paying off potential generic competitors not to enter the market, to keep the cash going even after the patents have all expired.
JGabriel
@patrick II:
It’s a little more complicated than that. If you have a side effect to a drug you need, than a “me too” drug that doesn’t have that side effect becomes the solution to an important problem. One person’s trivial difference is another’s life-saving change.
Let me add that I agree that fewer new drugs to get significantly cheaper prices, in a simultaneously over-priced yet over-saturated market, is probably the right choice – but we shouldn’t minimize or over-simplify the concerns of people who are unsure.
jl
@JGabriel: ” If you have a side effect to a drug you need, than a “me too” drug that doesn’t have that side effect becomes the solution to an important problem.”
I agree. But a critical issue is how difficult it is to solve those kinds of knock off problems. Importance to solve a problem for a particular patient population is different than difficulty of solving problem. As probability of success of research needed to solve problem gets closer to 100 percent, less excess profit from patent population is justified. Big returns from patent protection should be reserved for high risk products where chance of success is low. As noted above, there are many dimensions to how patent policy can be formulated and tweaked to get maximum social benefits out of it.
Edit: and note that in example given, we seem to have a patent that couples delivery device and the active drug. Plenty of examples from patent and approval process around the world the decouple the two, to a greater or lesser extent. US patent and approval system really emphasizes opportunity of excess profits from bundling the two.
PAM Dirac
@Roger Moore:
Are you sure the patent is extended in time? I thought doctrine of equivalents just extended the scope of the patent, it is a test for patent infringement, but if the patent has expired, infringement is no longer an issue.
Brachiator
@Roger Moore:
You left something out. Society loves drugs because they work.
Roger Moore
@PAM Dirac:
Of course how expensive a clinical trial is depends a lot on the size of effect you’re trying to detect and the kind of outcome you’re dealing with. If you’re developing a drug for a condition for which there’s currently no treatment, so you’re just comparing to placebo, and your drug really works, you may be able to show significant benefit with a relatively small patient cohort. If you’re trying to develop a competitor for an existing drug that’s expected to be marginally better, you need a huge number of patients to be able to show the size of benefit you’re expecting.
As an example, I’m peripherally involved* in a new drug application that I expect to have a very cheap Phase III trial. Everything is going their way. They’re treating an acute condition, so the course of treatment is short. Their Phase II trial showed a reduction in mortality from 20% to 7%, so there’s a big effect size. Also, since the clinical endpoint is death, they’re going to be relatively tolerant of side effects. They might need only a few hundred patients for their Phase III trial, and the whole thing is expected to take less than a year. Compare that to a clinical trial for a competitor in an area where there are already lots of existing drugs, like new statin. They might need to test thousands of patients for a really long time to have a chance to prove their drug works better than the best existing therapy.
*My lab does identity testing, so we are involved in testing the active ingredient to prove it is what it’s supposed to be.
Roger Moore
@PAM Dirac:
Because the formulation is a separate patent from the original patent on the active ingredient, it extends the time for which the company has an effective monopoly on their drug.
jl
@Brachiator: ” Society loves drugs because they work. ”
But when and how. Some research strongly suggests that US patent system tends to emphasize drug treatments that perferentially occur late in prognosis and emphasize, for noncancer drugs, maintenance and palliative effects rather than cure.
PAM Dirac
@Roger Moore:
I agree completely; there is a very wide range of conditions and numbers needed for a good clinical trial so there is a very wide range of clinical trial costs. But all clinical trials cost something and since “natural” remedies currently aren’t required to under go clinical trials and sold with just the suggestion that do anything useful at all, there is a huge disincentive to do any trials.
Mousebumples
Here is some background on how long drug patents last.
As a summary: 20 years. However, that’s from the moment the potential drug product is identified. Sure, PharmaCo can wait to apply for the patent on the chemical ingredient, but that risks a different company discovering the compound and getting their patent in place first. The studies (to prove safety, to prove efficacy) take time and definitely cut into that 20 year period before a drug is approved by the FDA.
Additional research (as outlined in the article linked) can extend the patent life. Did you find a new approved use for your drug? Have an extra 5 years. Did you reformulate your product into an Extended Release version? Get a new patent for your new product – and I think an extension on the existing patent too. I think they also offer extensions of patent life if studies are done in non-typical patient populations (e.g. children, pregnant women, etc.) since the typical preferred study population are healthy individuals from 20-40 or so.
Some of this is useful research that is definitely worth a patent extension. Some just feels like exploiting loopholes. I work for a pharmacy benefits manager (PBM) now, and so much of how our plan formulary is setup is to cover the cheaper generic meds and require trial and failure of the preferred options to get to the more expensive “me too” meds. (e.g. the newer strengths of fenofibrate that are way more expensive than the originally marketed strengths, Pexeva [paroxetine mesylate] versus generic Paxil [paroxetine hydrochloride], etc.) I’m not sure how much of that can be regulated, though, and I’m not sure if there’s an easy to solution to that. :/
Mousebumples
I’m in moderation, and i’m not quite sure what I said to put me in the penalty box. :(
Just Chuck
@PAM Dirac:
So a couple days of military spending.
PAM Dirac
@Roger Moore: @Roger Moore:
I’m not sure I understand what you mean by “effective monopoly”. I can’t see how you can get a new patent granted for something that would infringe an old patent*. So the company gets a patent on WonderDrug with formulation A. The doctrine of equivalents says that patent extends to all other formulations that give the same pharmacokinetics, efficacy, side effects, etc. Now the company patents WonderDrug with formulation B. I would expect that something in the pharmacokinetics, efficacy, side effects has to be different to get a new patent, although not necessarily different enough to be clinically relevant. The patent on formulation A expires and the company stops making it. In principle, formulation A is now generic and can be made by other suppliers. The question is is there a market for a reliable source of formulation A. It might be that formulation B is so much better clinically, that the answer is no. If so, I don’t really see a problem. On the other hand, if the answer is no because of payments that is a real problem that needs to be addressed. I also think that this is where the advertising (both to physicians and to potential patients) comes in. It might be that the answer is that there is no market because physicians have been essentially bribed or patients have been lead to believe that only the New Improved WonderDrug will cure their ills. That would be another serious problem and I suspect it contributes to the problem, but I’d like to see some data on how much of a contribution it makes and what it depends on.
*I actually have a patent on compounds intended to be potential drugs, so I had a lot of interaction with a patent lawyer, but mostly I found out that huge chunks of patent law don’t make much sense to scientists.
Mousebumples
@PAM Dirac:
My example response to this is completely anecdotal, but it illustrates a possible issue in the realm of mis-information.
Namenda (memantine) is a drug for Alzheimer’s/dementia that has been on the market for awhile. A few years ago (let’s say 2015, but that’s an estimate on my part), the drug company released Namenda XR, which is dosed 1 time per day versus 2 times per day – and cost a bunch more $$. A year or so later (2016 or 2017), I can’t seem to keep Namenda XR on my shelves, and my supplier runs into a backorder where they can’t keep it in stock either. I contacted a few MD offices about switching their patients _back_ to regular Namenda since I could order that to at least maintain their treatment with something. A few of the nurses informed me that they had been told by the drug company that regular Namenda was being discontinued, which was why they had changed all of their patients to Namenda XR.
Regular immediate-release Namenda was not being discontinued – but it was on the verge of losing it’s patent protection status. This trick (which might have just been by some nefarious drug reps in my neck of the woods) was intended to maximize the company’s ability to continue to profit off it’s brand-name protection by getting as many patients as possible to switch to Namenda XR versus continuing to take the older Namenda tablets.
In a vacuum, the existence of XR products is great. For patients that are prone to forgetfulness, needing to take less pills per day theoretically maximizes adherence to therapy. (though I seem to recall a study from when I was in pharmacy school a decade or so ago that showed that twice daily dosing typically had the best compliance of all potential treatment regimens – don’t ask me to find a cite/source, though) One daily dosing can also minimize one-off periods, though that is more of a concern for other disease states (e.g. Parkinson’s) than for Alzheimer’s.
However, in my pharmacy experience, patients typically go with whatever their doctor’s recommend and they don’t understand why such changes in therapy (which may simplify their pill-taking experience) may end up costing them (or their insurance company, or both of them) way more money. Cost sharing helps, theoretically, but when everything is expensive – especially in the hard to understand world of health care – it can be a challenge for patients to parse what added costs are worth it, and which ones aren’t.
Brachiator
@jl:
“Some research strongly suggests” really doesn’t mean very much. I would love to pick a couple of populous states and say, ban any drug that was created or patented after the year 2000, and observe what happens with respect to patient outcomes.
But otherwise it is damned hard to separate out the useful drugs from those that are less so. And I don’t know what to do with some of the suggestions here that innovation is really just greed and marketing bullshit.
Another Scott
@Brachiator: You’ve forgotten him, and Mylan’s EpiPen, already? It would be trivial to find other examples.
There are huge pressures in business to maximize profits. That too often means pushing the envelope to an extent that it would be criminal in a sane society.
Cheers,
Scott.
PAM Dirac
@Mousebumples: Thanks for that real world example. It does look like what I expected for the introduction of a new formulation; the new patent doesn’t prevent generics on the old patent from coming to market. From Wikipedia:
The problem really isn’t patent law, it is the various tricks, bribes, incentives, and marketing schemes that prevent or impede the production of a generic version that can complete with the “new improved” version.
Brachiator
@Another Scott:
The two questions are:
How do you promote innovation in medical care and treatment?
Do you reward innovation or presume that you can achieve similar results by models based on altruism or social good?
Zelma
What we are not underlining is the price gouging that occurs. I have a perfect example. I’m on a generic estrogen blocker. I started taking it in May. The “retail” price is almost $700 per month. With my Part D insurance, I pay about $80. I don’t know what my insurer really pays.
But I have some historical data which is very interesting. Five year ago, I was taking the same drug that was not a generic. I regularly hit the donut hole and had to pay full price. I don’t recall the exact amount, but I know it was less than $300 per month.
Now, explain to me why a generic now costs more than twice that amount? Obviously, the generic manufacturer did not bear the costs of research. And I’m relatively certain that the manufacturing costs are a small fraction of the price. Somebody is making a lot of money. It’s a racket.
Another Scott
@Brachiator: Dean Baker has written a lot of sensible pieces on these issues:
http://cepr.net/publications/briefings/testimony/drugs-are-cheap-why-do-we-let-governments-make-them-expensive
http://cepr.net/publications/reports/publicly-funded-clinical-trials-a-route-to-sustained-innovation-with-affordable-drugs-working-paper
Etc.
I’m not saying he has all the answers, but we have the drug-invention system we have because of choices we made. We can make different choices.
Cheers,
Scott.
Mousebumples
@PAM Dirac: Thanks for doing the Wiki searching. :) I didn’t realize that Actavis had really planned to pull the brand IR formulation from the market prior to the generic becoming available. I shouldn’t be surprised, however. >_>
These sort of tricks also makes me wonder if new drug variations (that would have a clinical significance) are intentionally delayed to maximize profits. I can think of a number of medications off the top of my head that come in various formulations. I’ve only been a practicing pharmacist for about a decade (*and I no longer work in the retail pharmacy), but I have to wonder how many of these sorts of things had the later iterations intentionally delayed so that it wasn’t competing against the original formulation.
Wellbutrin (buproprion) – IR (3x daily), SR (2x daily – also Zyban SR, which is “different” while still being basically the same, just for a different indication) and XL (1x daily)
Effexor (venlafaxine) – IR (2-3x daily), XR (1x daily)
Imitrex (sumatriptan) – tablets, injections, nasal spray
Zomig (zolmitriptan) – tablets, orally disintegrating tablets, and nasal spray
Namenda & Namenda XR (as above)
Fosamax (alendrone) – IR (daily) and weekly variations
Abilify (aripiprazole) – IR tablets (daily) and various longer acting injectables that have only come onto the market more recently (which are very helpful for patients with bipolar that may tend towards not taking their meds if they feel “fine”)
And I’m sure I’m missing plenty, but this is one of the things that makes me sad about the free market. No one else had a right to these drugs (due to the patent) – and there are probably laws (*even in the event of patent expiration) that would limit the ability of an outside company to market another company’s “intellectual property” under a new formulation, so skirting the patent laws to create a new formulation is probably not allowed. *sigh*
Brachiator
@Another Scott:
Thanks for this. I will take a look as time permits.
PAM Dirac
@Another Scott:
You are right that he is sensible and serious, but I think (at least in these papers) that he seriously underestimates how hard it will be to put the changes in place and how uncertain it will be that the system he describes will come even close to the current level of production (as weak as it is). I started writing a longer reply, but the thread is dead and there are more points to be made than fit in this format. I’ll bookmark the papers and try to bring up points as they come up in other threads.
TomatoQueen
@MattF: 1994, Thanks to Newtie and the Blowhards (Orrrin Hatch, who representated a state full of medical equipment, bogusceuticals, and big Pharma makers).